
Featured cases
UNiDTM Adaptive Spine Intelligence Case Library
Check back weekly for additional content featuring UNiDTM cases!
December 2022
L3-S1 fusion featuring UNiDTM ASI patient-specific rods
Patient description: 44-year-old female had previously underwent a laminectomy for a disc herniation. Patient presented with new onset of bilateral radicular leg pain without an antecedent history of trauma. Initial radiographic imaging showed laminectomy defect with degeneration at L4-5 and L5-S1. She initially underwent non-operative care, including PT/OT, steroid dose pack, and home stretching exercises. Pain continued and an MRI was ordered which showed significant foraminal stenosis at L5-S1 with facet arthropathy and significant disc degeneration. At the L3-4 and L4-5 levels, she was found to have facet arthropathy with disc height and foraminal collapse and further lateral recess/foraminal stenosis. She was also found to have a facet cyst at L4-5. Patient underwent transforaminal epidural steroid injections at the L4-5 and L5-S1 levels, which gave significant relief of her leg pain, but not her back pain. Her back pain was consistent with facet mediated symptoms. Of note, the patient had loss of lumbar lordosis with a PI-LL mismatch of 15 degrees. Her total lumbar lordosis was 44 degrees with a pelvic incidence of 58.
Patient diagnosis: multi-level lumbar degenerative disc disease at L3-S1, facet arthropathy with facetogenic low back pain L3-S1 (8/10 back), L4 and L5 bilateral radiculopathy (8/10 leg), and loss of lumbar lordosis with PI-LL mismatch.

Presented by:
Christopher Kleck, M.D.
University of Colorado Hospital
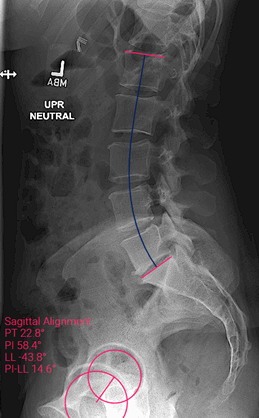
Sagittal pre-operative imaging
Case description: Patient underwent a L3-S1 anterior and posterior spinal fusion using UNiDTM patient-specific rods and the PASS® LP spinal system.
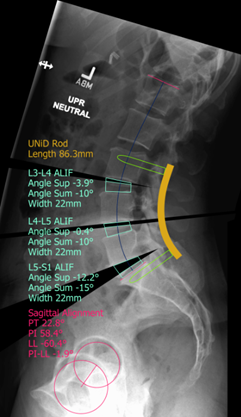
UNiDTM HUB Plan
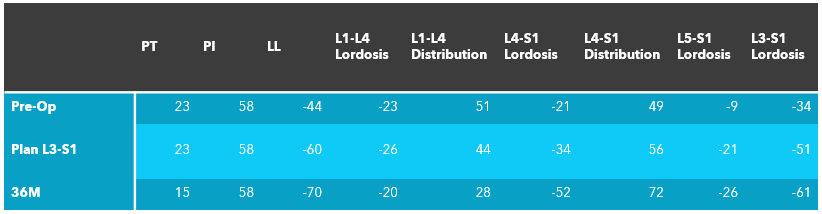
UNiDTM HUB Alignment Parameters
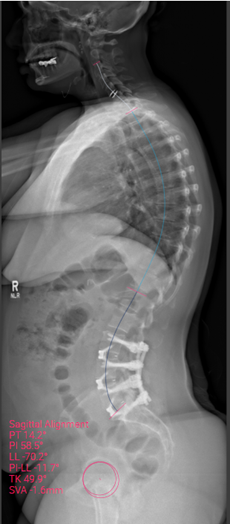
Sagittal postoperative image
Outcome: Radicular symptoms improved significantly after surgery. At the most recent post-op follow-up, patient reported no leg pain (0/10) and significant reduction in back pain (1/10 and occasional). Images and measurement show significant restoration of lumbar lordosis with correction of the pelvic tilt post-op.
Dr. Christopher Kleck is a paid consultant for Medtronic.
UNiD™ Spine Analyzer
The UNiD™ Spine Analyzer is intended for assisting healthcare professionals in viewing and measuring images as well as planning orthopedic surgeries. The device allows surgeons and service providers to perform generic, as well as spine related measurements on images, and to plan surgical procedures. The device also includes tools for measuring anatomical components for placement of surgical implants. Clinical judgment and experience are required to properly use the software.
The PASS LP® spinal systems
The PASS LP® spinal systems include a pedicle system intended to provide immobilization and stabilization of spinal segments in skeletally mature patients as an adjunct to fusion in the treatment of the following acute and chronic instabilities or deformities of thoracic, lumbar, and sacral spine:
Fractures.
Dislocation.
Failed previous fusion (pseudarthrosis).
Spinal stenosis.
Degenerative spondylolisthesis with objective evidence of neurological impairment.
Spinal deformations such as scoliosis or kyphosis.
Loss of stability due to tumors.
The PASS LP® spinal systems are also indicated for pedicle screw fixation for the treatment of severe spondylolisthesis (Grades 3 and 4) of the L5–S1 vertebra in skeletally mature patients receiving fusion by autogenous bone graft having implants attached to the lumbar and sacral spine (L3 to sacrum) with removal of the implants after the attainment of a solid fusion. The PASS LP® also include hooks and rods and sacral/iliac screws indicated for degenerative disc disease (ddd) defined as back pain of discogenic origin with degeneration of the disc confirmed by history and radiographic studies, spondylolisthesis, trauma (i.e., fracture or dislocation), spinal stenosis, deformities or curvatures (i.e., scoliosis, kyphosis, and/or lordosis), tumor, pseudarthrosis and failed previous fusion. Except for rod plates, when used for posterior non-cervical pedicle screw fixation in pediatric patients, the PASS LP® spinal system implants are indicated as an adjunct to fusion to treat adolescent idiopathic scoliosis. The PASS LP® spinal system is intended to be used with allograft and/or autograft. Pediatric pedicle screw fixation is limited to a posterior approach.
WARNING:
The safety and effectiveness of this device has not been established for use as part of a growing rod construct. This device is only intended to be used when definitive fusion is being performed at all instrumented levels.
RISKS:
In addition to the risks associated with surgery of the spine without instrumentation, a number of possible undesirable effects may occur with instrumented surgery (including but not limited to):
Detachment, deformation, mobilization, slipping, breakage of one or all of the components.
Pain due to the surgery, the fracture, deformation and or migration of an implant.
Fracture of the pedicle during insertion of a pedicular screw.
Postoperative loss of correction and/or reduction of the spine, partial or total loss of the corrections achieved.
CD Horizon™ Spinal System
The CD Horizon™ Spinal System with or without Sextant™ instrumentation is intended for posterior, non-cervical fixation as an adjunct to fusion for the following indications: degenerative disc disease (DDD - defined as back pain of discogenic origin with degeneration of the disc confirmed by history and radiographic studies), spondylolisthesis, trauma (i.e. fracture or dislocation), spinal stenosis, curvatures (i.e. scoliosis, kyphosis, or lordosis), tumor, pseudarthrosis, and/or failed previous fusion.
Except for hooks, when used as an anterolateral thoracic/lumbar system, the CD Horizon™ Spinal System titanium, cobalt chrome, and stainless steel implants may also be used for the same indications as an adjunct to fusion.
With the exception of DDD, CD Horizon™ Legacy™ 3.5mm rods and associated components may be used for indications in skeletally mature patients as an adjunct to fusion. The 3.5mm rods may be used for the specific pediatric indications noted. When used for posterior non-cervical pedicle screw fixation in pediatric patients, CD Horizon™ Spinal System titanium, cobalt chrome, and stainless steel implants are indicated as an adjunct to fusion to treat progressive spinal deformities (i.e. scoliosis, kyphosis, or lordosis) including idiopathic scoliosis, neuromuscular scoliosis, and congenital scoliosis. Additionally, the CD Horizon™ Spinal System is intended to treat pediatric patients diagnosed with the following conditions: spondylolisthesis/ spondylolysis, fracture caused by tumor and/or trauma, pseudarthrosis, and/or failed previous fusion. These devices are to be used with autograft and/or allograft. Pediatric pedicle screw fixation is limited to a posterior approach.
The CD Horizon™ PEEK rods are intended to provide posterior supplemental fixation when used with an interbody fusion cage for patients diagnosed with DDD. These DDD patients may also have up to Grade 1 spondylolisthesis or retrolisthesis at the involved level. This device is intended for 1-2 level use in the lumbosacral spine (L2 – S1) in skeletally mature patients. Devices are intended for use with an interbody fusion cage at the instrumented level and is not intended for stand-alone use.
The CD Horizon™ Spire™ plate is a posterior, single-level, non-pedicle supplemental fixation device intended for use in the non-cervical spine (T1-S1) as an adjunct to fusion in skeletally mature patients. It is intended for plate fixation/attachment to spinous processes for the purpose of achieving supplemental fixation in the following conditions: DDD, spondylolisthesis, trauma, and/or tumor.
To achieve additional levels of fixation, CD Horizon™ Spinal System rods may be connected to the Vertex™ Reconstruction System with the Vertex™ rod connector. Refer to the Vertex™ Reconstruction System package insert for a list of Vertex™ indications.
UC202308360EN CST
©2022 Medtronic. All rights reserved. Medtronic, Medtronic logo and Further, Together are trademarks of Medtronic. All other brands are trademarks of a Medtronic company.