
Featured cases
UNiDTM Adaptive Spine Intelligence Case Library
Check back regularly for additional content featuring UNiDTM cases!
June 2023
T4 - pelvis fusion featuring UNiDTM ASI patient-specific rods, O-armTM Surgical Imaging System, StealthStationTM navigation, and PowerEaseTM System
Patient description: 74-year-old female with a prior spinal fusion from T10-S1 performed in 2009. Patient continued to have chronic back pain and in 2019, she heard a "crack" and had worsening back pain and right buttock pain. Patient had increasing difficulty standing upright and could not walk long distances.
Patient diagnosis: Iatrogenic flatback deformity, pseudarthrosis with broken rods, proximal junctional failure

Presented by:
Venu Nemani, M.D.
Virginia Mason Franciscan Health (Seattle, WA)
Paid Consultant for Medtronic
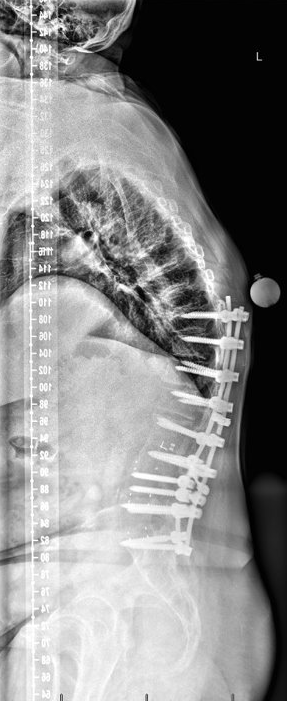
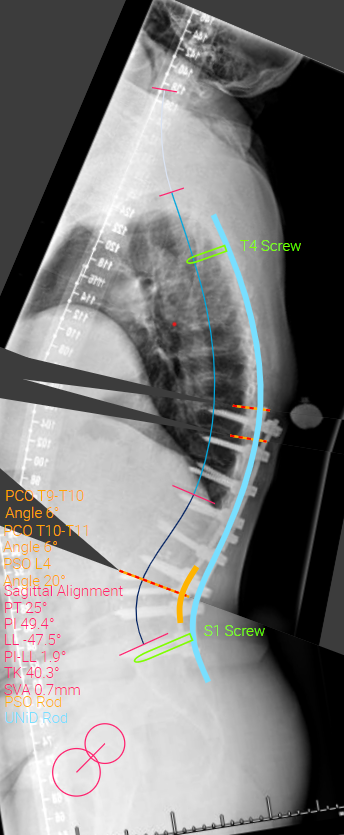
Saggital pre-operative imaging
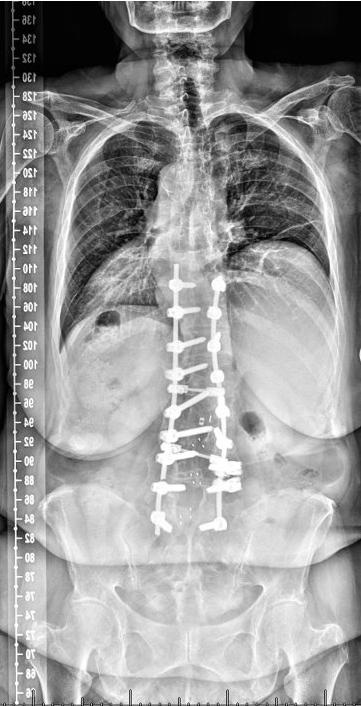
AP pre-operative imaging
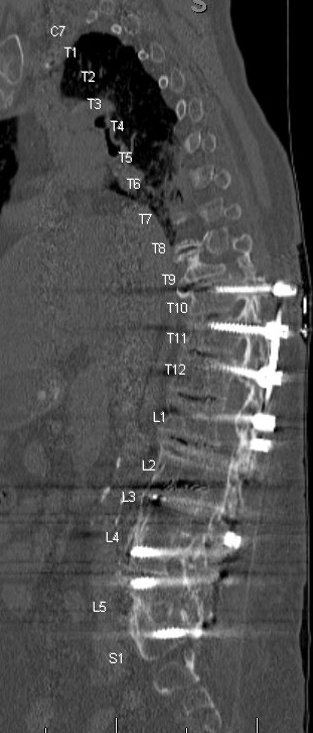
CT pre-operative imaging
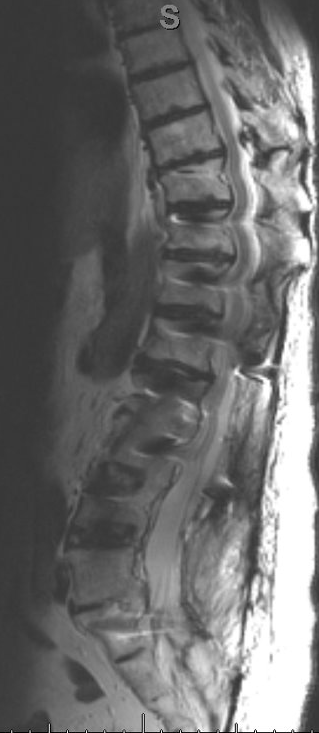
MRI pre-operative imaging
Case description: Patient underwent a revision posterior spinal fusion from T4 - pelvis with a pedicle subtraction osteotomy (PSO) at L4 and a tether from T2-T4. Screws were placed from T5 - T9 using 3D printed drill guides and PowerEase. T10-S1 was revised and instrumentation was upsized. Downgoing transverse process hooks were placed at T4. Stealth navigation was used to place bilateral S2AI screws and a third supplemental iliac screw. A PSO was performed at L4 and the osteotomy was closed using short rods spanning from L3-L5. Then, patient-specific rods were placed to realign the spine from T4 - pelvis. An additional rod was placed engaged into the supplemental iliac screw to have a 5 rods crossed the three-column osteotomy site. Finally, a tether was weaved in a figure-of-eight fashion through the spinous processes of T2-T4.
UNiDTM HUB Plan
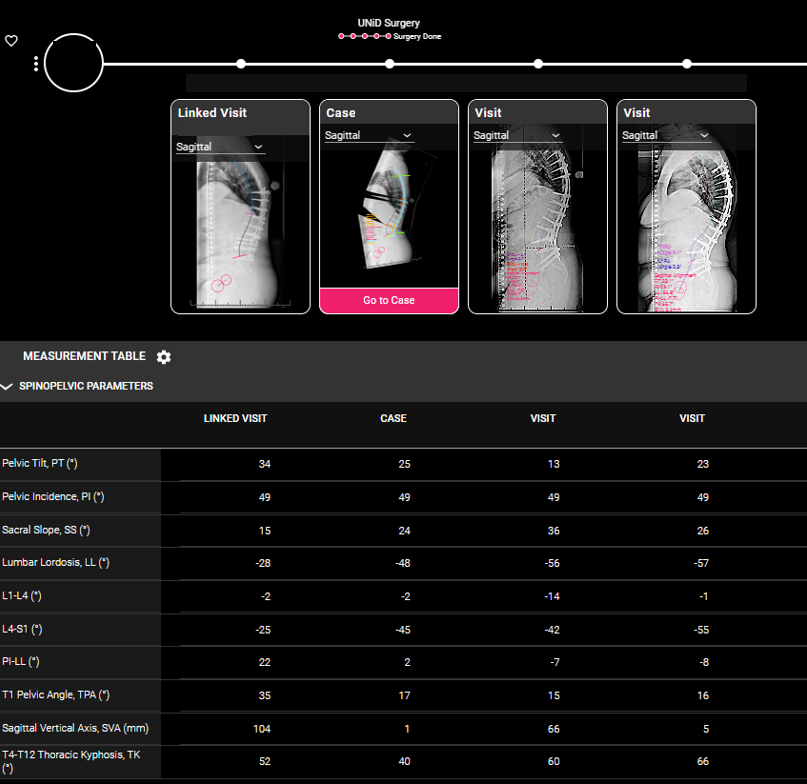
UNiDTM HUB Plan
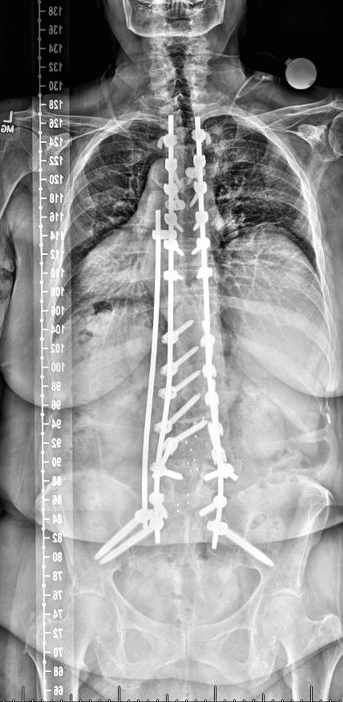
AP postoperative image
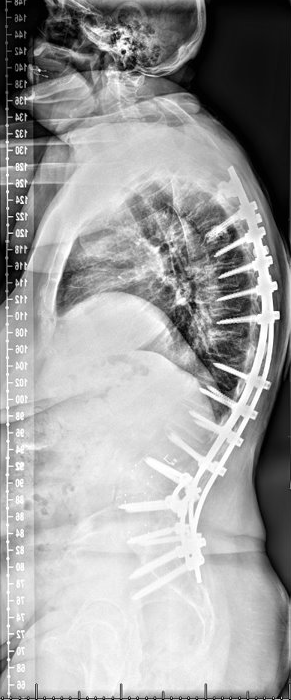
Saggital postoperative image
Outcome: Patient is 1-year postop with dramatic improvement in alignment, posture and reported improved pain compared to preop. ODI 38 preop, ODI 16 at 1-year postop.
Dr. Venu Nemani is a paid consultant for Medtronic.
UNiD™ Spine Analyzer
The UNiD™ Spine Analyzer is intended for assisting healthcare professionals in viewing and measuring images as well as planning orthopedic surgeries. The device allows surgeons and service providers to perform generic, as well as spine related measurements on images, and to plan surgical procedures. The device also includes tools for measuring anatomical components for placement of surgical implants. Clinical judgment and experience are required to properly use the software.
The PASS LPTM spinal systems
The PASS LPTM spinal systems include a pedicle system intended to provide immobilization and stabilization of spinal segments in skeletally mature patients as an adjunct to fusion in the treatment of the following acute and chronic instabilities or deformities of thoracic, lumbar, and sacral spine:
Fractures.
Dislocation.
Failed previous fusion (pseudarthrosis).
Spinal stenosis.
Degenerative spondylolisthesis with objective evidence of neurological impairment.
Spinal deformations such as scoliosis or kyphosis.
Loss of stability due to tumors.
The PASS LPTM spinal systems are also indicated for pedicle screw fixation for the treatment of severe spondylolisthesis (Grades 3 and 4) of the L5–S1 vertebra in skeletally mature patients receiving fusion by autogenous bone graft having implants attached to the lumbar and sacral spine (L3 to sacrum) with removal of the implants after the attainment of a solid fusion. The PASS LPTM also include hooks and rods and sacral/iliac screws indicated for degenerative disc disease (ddd) defined as back pain of discogenic origin with degeneration of the disc confirmed by history and radiographic studies, spondylolisthesis, trauma (i.e., fracture or dislocation), spinal stenosis, deformities or curvatures (i.e., scoliosis, kyphosis, and/or lordosis), tumor, pseudarthrosis and failed previous fusion. Except for rod plates, when used for posterior non-cervical pedicle screw fixation in pediatric patients, the PASS LPTM spinal system implants are indicated as an adjunct to fusion to treat adolescent idiopathic scoliosis. The PASS LPTM spinal system is intended to be used with allograft and/or autograft. Pediatric pedicle screw fixation is limited to a posterior approach.
WARNING:
The safety and effectiveness of this device has not been established for use as part of a growing rod construct. This device is only intended to be used when definitive fusion is being performed at all instrumented levels.
RISKS:
In addition to the risks associated with surgery of the spine without instrumentation, a number of possible undesirable effects may occur with instrumented surgery (including but not limited to):
Detachment, deformation, mobilization, slipping, breakage of one or all of the components.
Pain due to the surgery, the fracture, deformation and or migration of an implant.
Fracture of the pedicle during insertion of a pedicular screw.
Postoperative loss of correction and/or reduction of the spine, partial or total loss of the corrections achieved.
CD Horizon™ Spinal System
The CD Horizon™ Spinal System with or without Sextant™ instrumentation is intended for posterior, non-cervical fixation as an adjunct to fusion for the following indications: degenerative disc disease (DDD - defined as back pain of discogenic origin with degeneration of the disc confirmed by history and radiographic studies), spondylolisthesis, trauma (i.e. fracture or dislocation), spinal stenosis, curvatures (i.e. scoliosis, kyphosis, or lordosis), tumor, pseudarthrosis, and/or failed previous fusion.
Except for hooks, when used as an anterolateral thoracic/lumbar system, the CD Horizon™ Spinal System titanium, cobalt chrome, and stainless steel implants may also be used for the same indications as an adjunct to fusion.
With the exception of DDD, CD Horizon™ Legacy™ 3.5mm rods and associated components may be used for indications in skeletally mature patients as an adjunct to fusion. The 3.5mm rods may be used for the specific pediatric indications noted. When used for posterior non-cervical pedicle screw fixation in pediatric patients, CD Horizon™ Spinal System titanium, cobalt chrome, and stainless steel implants are indicated as an adjunct to fusion to treat progressive spinal deformities (i.e. scoliosis, kyphosis, or lordosis) including idiopathic scoliosis, neuromuscular scoliosis, and congenital scoliosis. Additionally, the CD Horizon™ Spinal System is intended to treat pediatric patients diagnosed with the following conditions: spondylolisthesis/ spondylolysis, fracture caused by tumor and/or trauma, pseudarthrosis, and/or failed previous fusion. These devices are to be used with autograft and/or allograft. Pediatric pedicle screw fixation is limited to a posterior approach.
The CD Horizon™ PEEK rods are intended to provide posterior supplemental fixation when used with an interbody fusion cage for patients diagnosed with DDD. These DDD patients may also have up to Grade 1 spondylolisthesis or retrolisthesis at the involved level. This device is intended for 1-2 level use in the lumbosacral spine (L2 – S1) in skeletally mature patients. Devices are intended for use with an interbody fusion cage at the instrumented level and is not intended for stand-alone use.
The CD Horizon™ Spire™ plate is a posterior, single-level, non-pedicle supplemental fixation device intended for use in the non-cervical spine (T1-S1) as an adjunct to fusion in skeletally mature patients. It is intended for plate fixation/attachment to spinous processes for the purpose of achieving supplemental fixation in the following conditions: DDD, spondylolisthesis, trauma, and/or tumor.
To achieve additional levels of fixation, CD Horizon™ Spinal System rods may be connected to the Vertex™ Reconstruction System with the Vertex™ rod connector. Refer to the Vertex™ Reconstruction System package insert for a list of Vertex™ indications.
StealthStation™ Surgical Navigation System
The StealthStation™ S8 System is intended as an aid for precisely locating anatomical structures in either open or percutaneous surgical procedures. The StealthStation™ System is indicated for any medical condition in which the use of stereotactic surgery may be appropriate, and where reference to a rigid anatomical structure, such as the skull, a long bone, or vertebra, can be identified relative to a CT or MR- based model, fluoroscopy images, or digitized landmarks of the anatomy.
The StealthStation™ S8 System with StealthStation™ Cranial software is intended as an aid for locating anatomical structures in either open or percutaneous procedures. Their use is indicated for any medical condition in which the use of stereotactic surgery may be appropriate, and where reference to a rigid anatomical structure, such as a skull, can be identified relative to images of the anatomy. This can include, but is not limited to, the following cranial procedures (including stereotactic frame-based and stereotactic frame alternatives-based procedures):
Tumor resections
Cranial biopsies
General ventricular catheter placement
Pediatric ventricular catheter placement
Depth electrode, lead, and probe placement
General localization
The StealthStation™ S8 System, with StealthStation™ S8 Spine Software, is intended as an aid for precisely locating anatomical structures in either open or percutaneous neurosurgical and orthopedic procedures. Their use is indicated for any medical condition in which the use of stereotactic surgery may be appropriate, and where reference to a rigid anatomical structure, such as the spine or pelvis, can be identified relative to images of the anatomy.
This can include the following spinal implant procedures, such as:
Pedicle Screw Placement
Iliosacral Screw Placement
Interbody Device Placement
CONTRAINDICATIONS:
Medical conditions which contraindicate the use of a Medtronic computer-assisted surgery system and its associated applications include any medical conditions which may contraindicate the medical procedure itself.
WARNINGS:
The system hardware and software should be used only by qualified medical professionals who are trained in performing surgery and familiar with image-guided surgery systems.
If system navigation seems inaccurate and recommended steps to restore accuracy are not successful, abort use of the system.
Inspect all visible system components before use including the localizers that accompany your system (that is, the camera, the EM instrument interface, the side-mount emitter, or the flat emitter). If visibly damaged, do not use the system.
To avoid the risk of electrical shock, do not simultaneously touch the patient and the system input/output panel, mouse, keyboard, or batteries. To avoid the risk of electric shock, this equipment must only be connected to a supply mains with protective earth.
Metallic and conductive objects in or near the electromagnetic navigation field can degrade navigational accuracy and the navigation volume. If metallic distortion causes excessive error, navigation may be disabled. To restore navigation, move metallic and conductive objects away from the navigation field.
Electrical noise in or near the electromagnetic navigation field can degrade navigational accuracy. If electrical noise introduces excessive error, the system may automatically disable navigation. To restore navigation, move devices that produce electrical noise (such as electrocautery equipment and electric drills) away from the navigation field.
Prevent fluid from entering any part of the StealthStation™ S8 system. Shut down the system, disconnect the power, and allow the system to dry if you suspect fluids may have entered any part of the system.
Do not step on the base or legs of the system carts because the carts could tip over and cause injury.
The system is not suitable for use in the presence of a flammable, anesthetic mixture with air, oxygen, or nitrous oxide.
Do not modify the StealthStation™ S8 system without prior written consent from Medtronic Navigation, Inc. Prohibited modifications include but are not limited to: altering, repairing, or replacing system components; altering software provided by Medtronic; updating the operating system or drivers; and installing any software that is not provided by Medtronic, including antivirus software. Secure the system to prevent unauthorized modifications. Unauthorized modifications could render the system unsafe or ineffective for its intended use.
Follow system component maintenance and care instructions.
The StealthStation™ S8 system camera cart contains a stereo system for music playback from a user provided audio player. This system may be capable of sound levels which can lead to permanent hearing loss at high volume. The volume is controlled at the user-provided audio player. For hearing safety, follow all instructions and warnings prescribed by the manufacturer of the user-provided player.
O™-arm Surgical Imaging System
The O-arm™ O2 Imaging System is a mobile x-ray system designed for 2D and 3D imaging for adult and pediatric patients weighing 60 lbs or greater and having an abdominal thickness greater than 16cm, and is intended to be used where a physician benefits from 2D and 3D information of anatomic structures and objects with high x-ray attenuation such as bony anatomy and metallic objects.
The O-arm O2 Imaging System is compatible with certain image guided surgery systems.
UC202307744EN CST
©2022 Medtronic. All rights reserved. Medtronic, Medtronic logo and Further, Together are trademarks of Medtronic. All other brands are trademarks of a Medtronic company.